
Rubric: Activity Deliverable Points Part I Complete Periodic Table simulation 10. assign observed wavelengths to correct Balmer transitions Why is hydrogen a particular theoretical interest? because it is the simplest of all atoms (only one proton and one electron) Who developed a theory for the hydrogen atom that explains the emission spectral lines as resulting from electron transitions between energy levels or discrete electron orbits? Niels Bohr what is the equation for the wavelengths of the spectral lines? wavelength= hc/deltaE What does the wave length of each emission spectrum depend on? the difference in energy as an electron changes from one energy level to another. View chem lab 2.docx from CHEM 120N at Chamberlain College of Nursing. 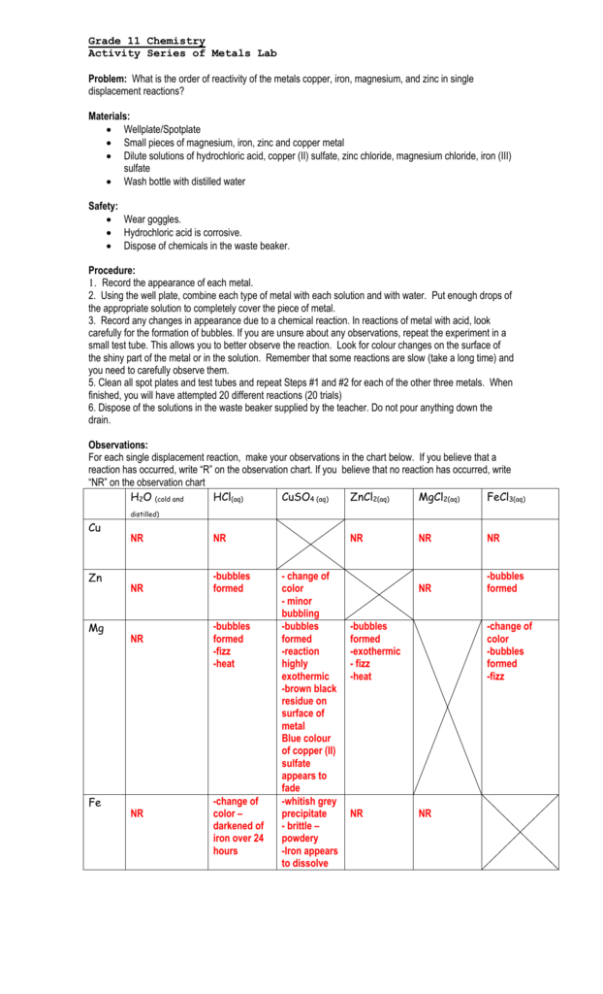
solve Rydberg equation using all four of possible Balmer transitionsĤ. The foliar applied silicon (Si) has the potential to ameliorate heavy metals, especially cadmium (Cd) toxicity however, Si dose optimization is strategically important for boosting the growth of soil microbes and Cd stress mitigation. In view of that the salt and complexes of rare earth metal have been tested by experiment and clinical use to possess antiinflammation, sterilization and antitumour activities 11, 12, 13, a. observe the spectrum for hydrogen and record three strongest color lines of hydrogen series (between blue, red, green, and violet)ģ. However, studies on Biological activities of the rare earth metal complexes with Curcumin and 1,10-phenanthroline-5,6-dione have been rarely reported. observe emission spectra for Argon, Neon, and helium gasses recording the four strongest lines seen in the spectraĢ. Clams were exposed to 0, 1, 3 and 10 for 7 and 14 day and the following biochemical. You will be looking for a plating reactiona darkening or coating of the metal’s surface. 158 2-6) Study Guide Section 6. 170) ASSESSMENT Section Assessment Questions (p. 155) CD Exploration: The Periodic Table Video: Transuranium Elements Demo: Activity of Alkali Metals Chemlab 6: Descriptive Chemistry of the Elements (p. If necessary, observe reactions through magnifying glasses. ACTIVITIES Discovery Lab: Versatile Metals (p. Can there be photons emitted outside of the visible light spectrum? Yes, but we can not see them What determines the size of the quantum jumps? the arrangement of electrons in an atom What does the emission spectrum serve as? a 'fingerprint' of the element to which the atoms belong How can you view the separate colors of an emission spectrum? by using a spectroscope What does a spectroscope do? it bends light of different energies differently In a spectroscope, the low energy (red light) is bent the _ most In a spectroscope, the high energy (violet light) is bent the _ least What happens when you see a rainbow? the water droplets in the air bend the colors of light so they appear as a spectrum What do astronomers use the emission spectrum for? to identify gases in the universe What will you do in part a of this lab? we will do several flame tests that focus on emission spectra List the steps of Part A in this experiment. you do not need to test free metal A in the metal cation A solution because it won’t react with itself.) Allow each metal to react with each solution for at least 10-15 minutes.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
